News
Technical guides, equipment comparisons, and compliance insights for pharmaceutical manufacturing professionals.

Continuous or Traditional? Building an Efficien...
Traditional ODF manufacturing often uses stand-alone machines and experience-based scale-up, which can struggle with complex films and high validation costs. This article compares traditional and integrated continuous models, and explains...
Continuous or Traditional? Building an Efficien...
Traditional ODF manufacturing often uses stand-alone machines and experience-based scale-up, which can struggle with complex films and high validation costs. This article compares traditional and integrated continuous models, and explains...

ODF Line Validation Checklist: From URS to FAT,...
This article outlines a practical validation checklist for ODF production lines—from URS and FAT/SAT through IQ/OQ/PQ. It shows how to treat the line as one system rather than isolated machines,...
ODF Line Validation Checklist: From URS to FAT,...
This article outlines a practical validation checklist for ODF production lines—from URS and FAT/SAT through IQ/OQ/PQ. It shows how to treat the line as one system rather than isolated machines,...

From Slitting to Single Sachets: Traceability a...
For ODF products, the packaging section turns coated film into market-ready, auditable units. This article explains how the MJF180 integrates slitting, sealing, traceability points, and moisture control to protect dose...
From Slitting to Single Sachets: Traceability a...
For ODF products, the packaging section turns coated film into market-ready, auditable units. This article explains how the MJF180 integrates slitting, sealing, traceability points, and moisture control to protect dose...

Multi-Formula Stripe Coating Process Window: Vi...
Learn how to optimize the multi-formula stripe coating process in ODF production. By carefully managing viscosity, leveling, and drying, you can achieve consistent results and flexibility in formulation combinations.
Multi-Formula Stripe Coating Process Window: Vi...
Learn how to optimize the multi-formula stripe coating process in ODF production. By carefully managing viscosity, leveling, and drying, you can achieve consistent results and flexibility in formulation combinations.

Line Selection Guide: How MJ150-L and MJ150 Wor...
Choosing between MJ150-L and MJ150 is not only about capacity. This guide explains how to start with MJ150-L for formulation and SOP definition, when to migrate to MJ150, and how...
Line Selection Guide: How MJ150-L and MJ150 Wor...
Choosing between MJ150-L and MJ150 is not only about capacity. This guide explains how to start with MJ150-L for formulation and SOP definition, when to migrate to MJ150, and how...

Vitamin D Oral Dissolving Film (ODF): From Coat...
This case study walks through a production-ready ODF route for Vitamin D — starting at precision coating and drying, through slitting/die-cutting, and into single-sachet packaging. It excludes liquid preparation and interfaces...
Vitamin D Oral Dissolving Film (ODF): From Coat...
This case study walks through a production-ready ODF route for Vitamin D — starting at precision coating and drying, through slitting/die-cutting, and into single-sachet packaging. It excludes liquid preparation and interfaces...

CIPM 2025 Qingdao Recap | HUANGHAI Highlights i...
Our CIPM 2025 Qingdao recap: modular ODF line from lab to commercial, laser drilling with inspection and data integrity, and QC instruments for GMP workflows.
CIPM 2025 Qingdao Recap | HUANGHAI Highlights i...
Our CIPM 2025 Qingdao recap: modular ODF line from lab to commercial, laser drilling with inspection and data integrity, and QC instruments for GMP workflows.
How ODF Coating Lines Meet FDA 21 CFR Part 11 C...
As ODF dosage forms expand globally, regulatory compliance has become a key evaluation criterion for pharmaceutical manufacturers.HUANGHAI’s MJ150 and MJ150-L ODF coating systems are designed with full FDA 21 CFR...
How ODF Coating Lines Meet FDA 21 CFR Part 11 C...
As ODF dosage forms expand globally, regulatory compliance has become a key evaluation criterion for pharmaceutical manufacturers.HUANGHAI’s MJ150 and MJ150-L ODF coating systems are designed with full FDA 21 CFR...
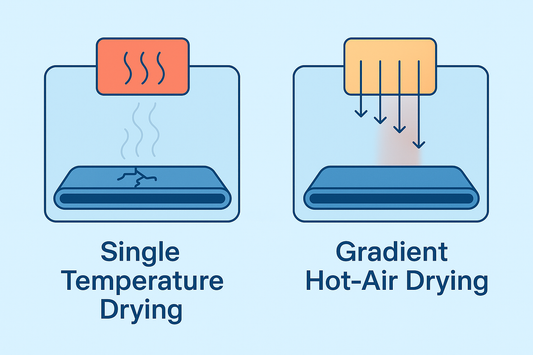
Gradient Hot-Air Drying: The Key to ODF Yield a...
Efficient drying determines the quality and yield of ODFs and transdermal patches. HUANGHAI’s patented gradient hot-air drying system prevents cracks, curling, and solvent residue by gradually increasing temperature with natural...
Gradient Hot-Air Drying: The Key to ODF Yield a...
Efficient drying determines the quality and yield of ODFs and transdermal patches. HUANGHAI’s patented gradient hot-air drying system prevents cracks, curling, and solvent residue by gradually increasing temperature with natural...
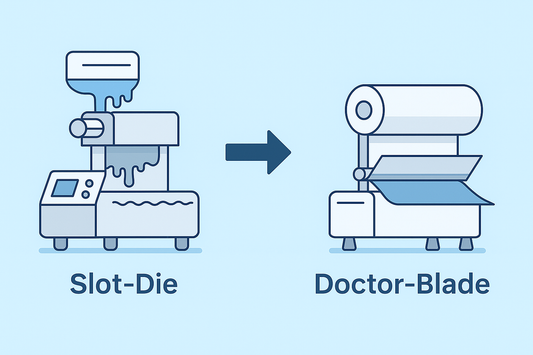
Doctor-Blade vs Slot-Die: Choosing the Right Co...
Coating method determines uniformity, stability, and scalability for ODFs and transdermal films. This article compares doctor-blade and slot-die, explains common failure modes with high-viscosity formulations, and shows why HUANGHAI adopts...
Doctor-Blade vs Slot-Die: Choosing the Right Co...
Coating method determines uniformity, stability, and scalability for ODFs and transdermal films. This article compares doctor-blade and slot-die, explains common failure modes with high-viscosity formulations, and shows why HUANGHAI adopts...
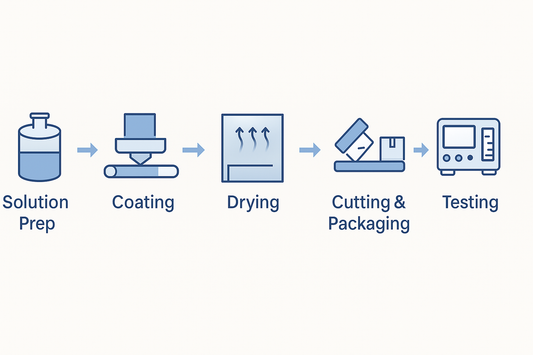
How to Build a Modular ODF Production Line for ...
A practical blueprint for configuring a modular ODF production line that adapts to very different dosage strengths—from microgram to tens of milligrams. See how to mix-and-match coating, drying, cutting/packaging, and...
How to Build a Modular ODF Production Line for ...
A practical blueprint for configuring a modular ODF production line that adapts to very different dosage strengths—from microgram to tens of milligrams. See how to mix-and-match coating, drying, cutting/packaging, and...
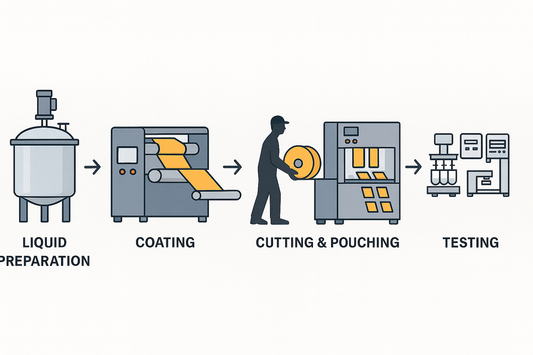
Configuring Pharma-Grade ODF Production Lines: ...
Pharma-grade ODF production requires more than coating and packaging. A closed-loop system—from liquid prep to QC—ensures GMP compliance, traceability, and registration-ready data for global markets.
Configuring Pharma-Grade ODF Production Lines: ...
Pharma-grade ODF production requires more than coating and packaging. A closed-loop system—from liquid prep to QC—ensures GMP compliance, traceability, and registration-ready data for global markets.
