Patented Hot-Air Drying: Boosting ODF & Transdermal Patch Efficiency by 30%
Share
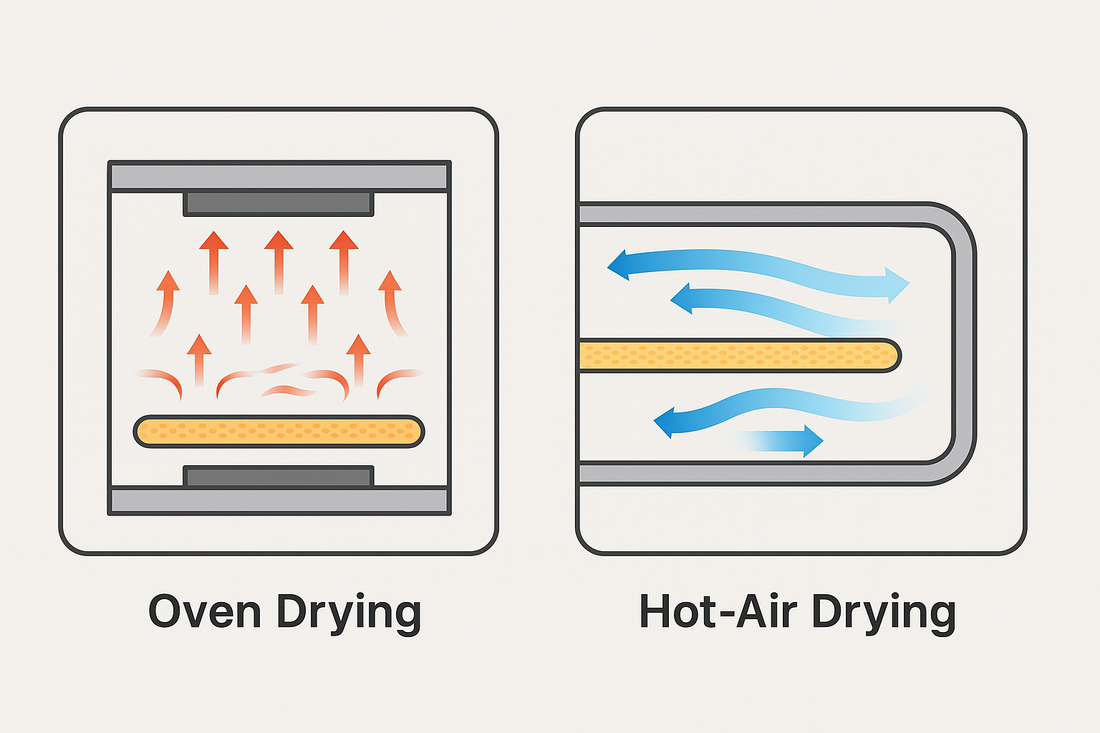
In the large-scale production of oral dissolvable films (ODFs) and solvent-based transdermal patches, the drying process is often the critical bottleneck. Traditional oven-type heating systems, which rely on top and bottom heating plates, frequently struggle to deliver the required efficiency, uniformity, and scalability demanded by modern pharmaceutical manufacturing. To overcome these challenges, Huanghai has developed an exclusive patented hot-air drying technology (CN201668734U), designed specifically for pharmaceutical-grade film production lines.
Industry Pain Points: Why Drying Technology Matters
- Uneven heating with oven plates: Conventional oven heating creates hot spots and temperature fluctuations, often leading to inconsistent film quality.
- Low throughput: Oven-type systems have limited drying efficiency, resulting in longer production cycles and higher operational costs.
- Lack of progressive temperature control: ODFs and patches require a smooth temperature gradient during drying. Oven systems can only achieve stepwise heating changes, requiring frequent manual adjustments.
- Scaling challenges: When moving from pilot-scale trials to full commercial production, oven-based drying fails to maintain consistent performance at higher outputs.
The Huanghai Solution: Patented Hot-Air Drying Technology
Huanghai’s exclusive hot-air drying system (CN201668734U) is engineered to address these challenges head-on. Integrated into the MJ150 ODF & Transdermal Patch Film Making Machine, this patented technology delivers superior performance in large-scale pharmaceutical applications.
Key advantages include:
- Higher heating efficiency & uniformity: Hot-air circulation provides consistent temperature distribution, reducing defects and ensuring stable product quality.
- Progressive temperature control: Supports a smooth transition from hot to mild conditions, protecting sensitive drug formulations and maintaining film integrity.
- 20–30% increase in drying efficiency: Compared to oven-type heating, Huanghai’s system significantly boosts output capacity while lowering energy costs.
- Optional far-infrared heating: Additional modules can be integrated to further enhance total throughput.
- Exclusive competitive edge: This patented system is not available in competing solutions such as oven-type systems from other suppliers, ensuring Huanghai customers benefit from unique performance advantages.
Conclusion
As the pharmaceutical industry continues to demand faster, more reliable, and scalable production of ODFs and transdermal patches, drying technology has become the defining factor for efficiency and compliance. With its patented hot-air drying system, Huanghai provides manufacturers with a powerful tool to achieve higher throughput, superior film quality, and full regulatory compliance.
Discover more about our large-scale MJ150 Film Coating Machine, or contact our team to explore how Huanghai can support your ODF and patch manufacturing needs.
*Features Huanghai's exclusive patented hot-air drying technology (Patent No. CN201668734U) ensuring uniform bottom-up heating.
*Designed to meet Class I Div 1 / ATEX explosion-proof standards for safe transdermal solvent handling.
Frequently Asked Questions
Q: What is the difference between an ODF film and a transdermal patch?
A: Oral Dissolving Films (ODF) are placed on or under the tongue and dissolve within seconds to minutes, delivering APIs directly through the oral mucosa or via swallowing. Transdermal patches adhere to the skin and deliver APIs through the dermal layers into systemic circulation over hours to days. Despite different delivery routes, both are manufactured using similar solvent-cast film coating processes. Huanghai's MJ150 ODF machine supports both applications on a single platform with approximately 2 working days of changeover time.
Q: What production output can I expect from a Huanghai ODF machine?
A: The MJ150 produces 20,000 films/hour at commercial scale. The MJ150-L targets R&D and pilot production at 8,000–10,000 films/hour. For fully integrated lines, pair either machine with the MJF180 automatic cutting and packaging system (11,900 films/hr) or the more affordable EZ320 (9,000 films/hr). A complete MJ150 + MJF180 line can produce over 150 million finished pouches annually on a single-shift basis. Contact us for pricing and configuration details.
Q: What drying technology does Huanghai use for ODF production?
A: Huanghai uses patented gradient hot-air drying (Patent CN201668734U), which applies a smooth progressive temperature drop rather than stepwise oven zones used by competitors. This results in more uniform film thickness, reduced edge curl, and better API distribution across the film web. An optional far-infrared heating module adds 20–30% drying efficiency for solvent-based formulations. This patented drying system is one of the key technical advantages that justifies Huanghai's position as the preferred ODF equipment supplier to Sinopharm, Shanghai Pharma, and Fosun Pharma.
Q: What are the GMP requirements for ODF production equipment?
A: ODF manufacturing equipment must comply with cGMP (21 CFR Parts 210/211) for US market products, and equivalent standards (EU GMP Annex 1, ChGMP) for other markets. Key requirements: material contact surfaces must be 316L stainless steel or equivalent; CIP/SIP capability or documented cleaning validation; data integrity controls meeting ALCOA+ principles (audit trails, access control). Huanghai ODF machines meet these standards—all product contact surfaces use pharmaceutical-grade materials, and the control system includes operator access logs and parameter change records. Request our GMP compliance documentation.
Q: Can Huanghai machines produce stripe-coated or multi-formula ODF films?
A: Yes. Huanghai holds a patent for multi-formula stripe coating (Patent CN117323228A), enabling two different API formulations to be applied side-by-side in a single coating pass. This eliminates the need for multiple coating/drying cycles when producing combination-drug ODF products. Competitors require manual multi-layer coating with drying intervals between each formula. This capability is particularly valuable for fixed-dose combination products (e.g., dual-API ODFs for cardiovascular or CNS indications) where coating efficiency directly impacts production economics.
